Environmental Sensitivity and Serotonin Transporter Gene Variance: from Rats to Humans
27th March 2020 - By Judith R Homberg
About the authors
Judith Homberg is professor Translational Neuroscience at the Donders Institute for Brain, Cognition, and Behaviour in Nijmegen, The Netherlands. Her preclinical research group focusses on understanding individual differences in behavior in relation to vulnerability and resilience to neurodevelopmental and stress-related disorders.
Summary
Environmental Sensitivity is seen in both animals and humans, suggesting that is has a strong biological foundation. One biological factor that increases environmental sensitivity is the serotonin transporter gene. This study demonstrated that environmental sensitivity is associated with similar neural substrates in humans and animals with inherited serotonin transporter down-regulation.
Background information
Environmental sensitivity is thought to have its roots in evolution. Indeed, it is pertinent for the overall stability of many animal populations, including human populations, that there are individuals who are bold and little influenced by the environment as well as individuals who are cautious and highly sensitive to the environment. Such an evolutionary basis suggests that individual differences in environmental sensitivity have a strong biological foundation.
Genetic Differences in Sensitivity
One exemplary gene variance that confers environmental sensitivity is the serotonin transporter linked polymorphic region (5-HTTLPR), which describes a section on the DNA that comes in either a short or a long version, referred to as allelic variants The short allelic variant, compared to the long allelic variant, of this promotor gene variance is associated with reduced expression of the serotonin transporter (5-HTT) and increased environmental sensitivity.
This gene variance is found in humans and non-human primates, but not in other animal species. Because of species differences in the promotor region of the 5-HTT gene, genetic engineering attempts to induce the 5-HTTLPR in other animal species failed. Nonetheless, it can be modelled in rodents through 5-HTT knockout, thus inactivation of the 5-HTT gene.
While a gene knockout is not an exact copy of a promotor polymorphism, there is striking overlap in the behavioural effects of the polymorphism in humans and non-human primates and the gene knockout in rodents.
Thus, humans, non-human primates and serotonin transporter knockout mice and rats all demonstrate increased cautiousness and anxiety, and increased responsivity to both aversive and rewarding environmental stimuli.
In humans these can be pictures of sad and happy faces, and in animals stress (e.g. social defeat) and reward (e.g. sucrose, environmental enrichment) [1, 2]. This suggests that the function of the serotonin transporter is highly conserved and that its effects on behaviour are mediated through similar neural mechanisms (i.e., genetic effects influences behaviour through specific neural mechanisms). However, this has not been empirically tested.
Study Aims
The aim of our study [2] was to examine whether environmental sensitivity in humans carrying the serotonin transporter promotor polymorphism and rats lacking the serotonin transporter are mediated through similar neurocircuitries.
How the human study was conducted
We focused on sensitivity to a negative stimulus (threat), thus one side of environmental sensitivity, because threat sensitivity can be tested in humans and rats alike. We tested healthy human volunteers (N=104) in a probabilistic fear conditioning paradigm (i.e., an experiment in which participants learned to pair a stimulus with a negative experience), which was conducted in a brain scanner (MRI) and combined with heart rate measurements using an oxymeter.
Humans received electric shocks through a finger index, while watching simple cues presented on a screen. One of the cues predicted the shocks with a probability of 30%. We specifically determined the physiological and associated neural response to threat predicting cues in the absence of the threat (electric shock) itself.
Key findings
We found that in response to threat individuals carrying the 5-HTTLPR short allelic variant displayed increased fear bradycardia (a reduction in heart rate) and that this was mediated by increased connectivity between the amygdala (located deep in the brain and ‘signaling’ emotion) and the periaqueductal gray (located in the brain stem and mediating autonomic responses such as heart rate).
How the animal study was conducted
To determine whether 5-HTT knockout rats would display similar physiological and neural correlates, we subjected 5-HTT knockout and control rats to a fear conditioning and extinction paradigm.
During fear conditioning auditory tones were associated with footshocks. During fear extinction the tones were presented without footshock and the behavioural and physiological responses were measured.
Key findings
We observed that 5-HTT knockout rats displayed increased freezing (no movement except for breathing) and fear bradycardia, like in human 5-HTTLPR short allele carriers. The freezing may reflect ‘pause-to-check’ as is typically observed in cautious individuals.
To study whether the freezing and fear bradycardia were in 5-HTT knockout rats also mediated by altered connectivity between the amygdala and periaqueductal gray, we conduced ex vivo immunostainings, focusing on GABAergic somatostatin positive neurons in the amygdala known to project to the periaqueductal gray.
The activity of these neurons was in increased in the 5-HTT knockout and found to significantly mediate the association between the 5-HTT gene and freezing [3].
General conclusions
This study demonstrates that behavioural and physiological similarities between human 5-HTTLPR short allele carriers and 5-HTT knockout rodents have similar neural correlates, making it tentative to use the rodents to further understand the neurobiological basis of environmental sensitivity.
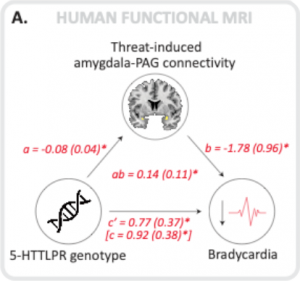
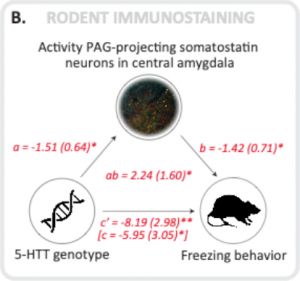
A mediation analysis shows that both in humans and in rats the connectivity between the amygdala and the periaqueductal gray (PAG) significantly mediates the association between inherited serotonin transporter (5-HTT) gene variance and fear bradycardia (humans) and freezing rats.
References
- Homberg, J.R. and K.P. Lesch, Looking on the bright side of serotonin transporter gene variation. Biol Psychiatry, 2011. 69(6): p. 513-9.
- Homberg, J., et al., Sensory processing sensitivity and serotonin gene variance: Insights into mechanisms shaping environmental sensitivity. . Neurosci Biobehav Rev, 2016. 71: p. 472-483.
- Schipper, P., et al., The association between serotonin transporter availability and the neural correlates of fear bradycardia. Proc Natl Acad Sci U S A, 2019. 116(51): p. 25941-25947.
